Accomplishments
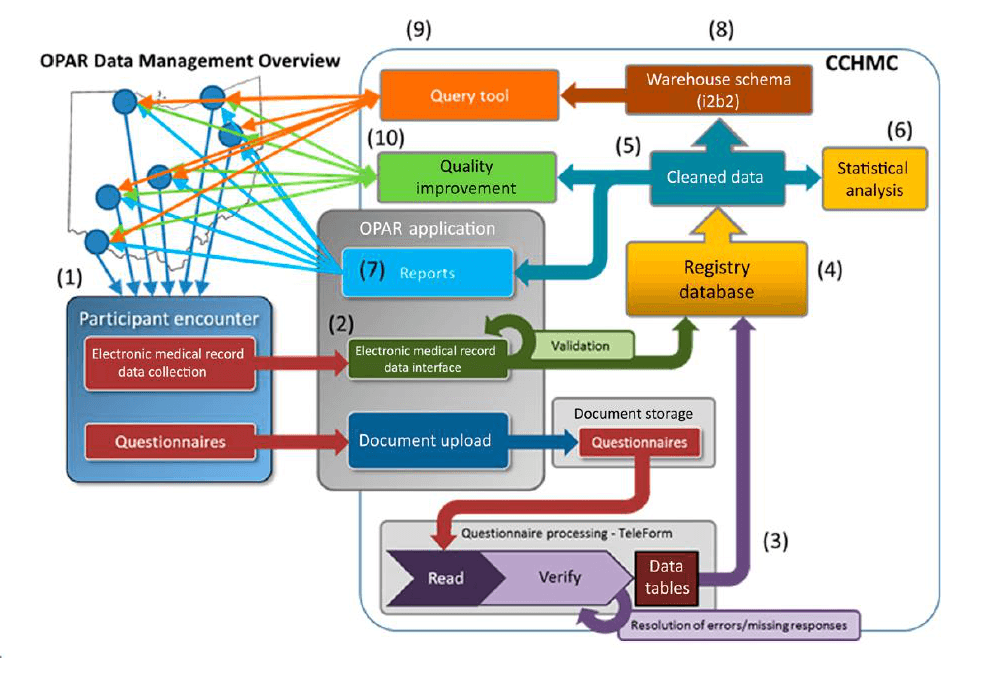
The OPAR study manages and analyses data collected from participants at all participating hospitals through the use of a centralized web-based tracking system, and makes data available for query via i2b2.
Central IRB
The OPAR study was an early adopter of a new IRB reliance agreement in place across many Ohio academic institutions. Using this reliance agreement, and creating reliance agreements for other sites, OPAR was able to establish a Central IRB whereby participating site IRBs agree to rely on the Cincinnati Children’s Hospital Medical Center IRB. This agreement helped the OPAR study get up and running more quickly, protocol amendments to be approved and implemented at participating sites in a timely and uniform manner, and reduced the regulatory burden on many of the participating sites.
i2b2
i2b2 (Informatics for Integrating Biology & Bedside) is a data storage and sharing tool that allows each OPAR site to create and run custom data queries on OPAR data. Using i2b2, OPAR staff can create queries with specific criteria, and determine how many study participants fit that criteria. In turn, this allows OPAR staff to see whether there is a cohort to fit their need, and if that cohort would provide enough statistical power to answer their research questions.
Publications
- Biagini Myers JM, Simmons JM, Kercsmar CM, Martin LJ, Pilipenko VV, Austin SR, Lindsey MA, Amalfitano KM, Guilbert TW, McCoy KS, Forbis SG, McBride JT, Ross KR, Vauthy PA, Khurana Hershey GK. Heterogeneity in Asthma Care in a Statewide Collaborative: the Ohio Pediatric Asthma Repository. Pediatrics. 2015 Jan 19. pii: peds.2014-2230. [Epub ahead of print]
- Biagini Myers JM, Austin S, Lindsey MA, Martin LJ, Pilipenko VV, Kercsmar CM, Simmons JM, Guilbert TW, McCoy K, Forbis S, McBride J, Ross K, Vauthy PA, Khurana Hershey GK. A comparison of clinical practices at six Ohio Children’s Hospitals: The Ohio Pediatric Asthma Repository (OPAR). Am J Crit Care Med. 2014. 189; 204:A1825